  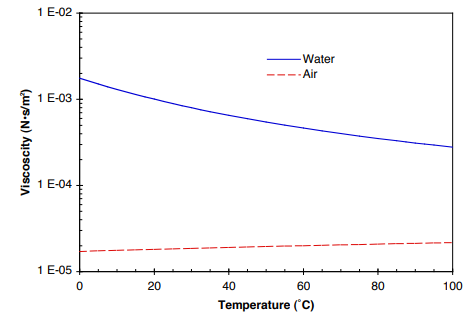
Now, if you came back to that baking sheet later, the honey might have spread out across the baking sheet.īut that difference between water and honey, and the time it took to move across that plane is dynamic viscosity.ĭynamic viscocity is not to be confused with kinematic viscocity. It will mostly retain the shape it was in when it came in contact with the shape. Its internal friction is just too low to keep its own shape (unless it's frozen). Water, since it has a low viscosity, it will likely spread out all across the baking sheet. To illustrate this, imagine pouring a cup of water over a metal baking sheet. Specifically, it's a measure of liquid's resistance to move across a horizontal plane. It is defined as, "the force needed by a fluid to overcome its own internal molecular friction so that the fluid will flow." The Ideal Gas Law - The relationship between volume, pressure, temperature and quantity of a gas, including definition of gas density. 
Sulfur Dioxide Liquid - Thermal Properties - Density, specific heat, thermal conductivity and more. Now that we know what viscosity is, let's move on to dynamic viscosity. Pressure - Absolute viscosity of steam at pressure ranging 1 - 10000 psia. If you were to flip a cup of honey upside down, you might be there a while before it fully leaves the cup. So if you were to flip a cup of water upside down, the water would spill everywhere immediately. It is relatively frictionless when compared to a liquid like honey or molasses, which are both extremely viscous. Water has a very low viscosity because it flows easily and freely. Viscosity is a trait or measure of a liquid that has resistance to "deformation" or movement when put under stress, whether that's gravity or some other force. To answer this question, it is probably easiest to first define what viscosity is. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
